
X-Ray Crystallography
About

The X-ray Crystallography facility offers X-ray diffraction services for both single crystal and powder/polycrystalline materials. The facility is well-equipped to investigate many different types of materials from different research fields.
We offer a complete service for single crystal structure solution and refinement of small molecules. The Cambridge Structural Database (from the CCDC), the PDF-2 database (from the ICDD), and a variety of software programs for structural analysis and processing are available for researchers to use at various software stations.
Contact:
For X-ray crystallographic services please contact:
Brian Patrick, Ph.D
Tel: (604)-822-4865
Fax: (604)-822-2847
E-mail: bpatrick@chem.ubc.ca
Our Members:
Dr. Brian Patrick, Manager - bpatrick@chem.ubc.ca
Anita Lam (PXRD specialist) - aelam@chem.ubc.ca
Location:
We are located in Chemistry – North wing, rooms E214, E215, and E217 (office).
Instruments
Bruker D8 Venture
- Two high intensity DIAMOND II microsources (Ag and Cu)
- Photon III Area Detector (14 x 10 cm2)
- Oxford Cryostream 1000 (80K – 400K range)
- Kappa geometry
- Equipped with Bruker’s APEX single crystal structure determination software
- Equipped with Bruker’s DIFFRAC. EVA and TOPAS software for PXRD (including PDF analyses) and materials XRD applications

Bruker Apex DUO
- Two X-ray sources: Mo and Cu
- Greater Mo intensity with the Triumph monochromator - useful for challenging or weakly diffracting crystals
- High intensity Cu microsource radiation for the determination of absolute structures for light atom organic compounds
- Automatic data collection strategy determined using COSMO, cell determination and integration carried out with SAINT (or CELL_NOW for twinned crystals)
- Kappa geometry, allows for crystal orientation

Malvern-Panalytical Empyrean 3
The Empyrean 3 is a multi-purpose diffractometer with many functionalities.
- BBHD mirror
- iCore and dCore modules – fast and automated optical configuration changes
- PIXcel 3D detector – 0D, 1D, 2D modes
- Theta-theta goniometer for Bragg-Brentano, transmission, grazing incidence and other configurations
- Stages: reflection/transmission spinner (use with automatic sample changer), basic, and micro-diffraction
- Software: HighscorePlus, DataViewer, XRD2DScan, PANViewer, Stress (basic), AMASS (basic)
Bruker D8-Advance
- Bragg-Brentano configuration
- For routine data collection and identification of powder and materials
- Cu source with LynxEye silicon strip detector
- 9-position sample changer
- Bruker EVA and TOPAS software

Bruker D8-Discover
- Bragg-Brentano configuration with point detector
- TTK450 temperature stage for in-situ variable temperature experiments (inquire about availability)
Note: Variable Temperature experiments also available with the DUO SCXRD

Services
Single Crystal Links:
Click here to view OLEX2 Cheat Sheet (Adobe PDF format).
Click here to view Ilia Guzei notes on OLEX2 (Adobe PDF format).
Sample submission - Single Crystal XRD:
Click here toprint a Submission Form (Adobe PDF format).
Click here to view a sample of a completed submission form (Adobe PDF format).
How to keep the Crystallographer happy; some general rules and advice:
Please note: It has always been the policy of this facility that the crystallographer is included as an author in any paper or publication where the results of the single crystal structure solution are presented.

- Avoid submitting a polycrystalline mass of solid material. The sample should consist of carefully grown single crystals. A crystal is defined by straight edges and plane faces (see any crystallography text for further information). If your sample does not have the prerequisite morphology, the crystallographer would be happy to look at it, but keep in mind that you may have to recrystallize it.
Crystals used in the diffraction experiment are typically no larger than 0.5 x 0.5 x 0.5 mm in each direction. X-ray quality crystals that are larger in dimension are cut by the crystallographer to a suitable size. Very small crystals can also be submitted to the crystallographer, but these samples may be too weakly scattering. In such cases, recrystallization to obtain larger crystals may be necessary if the diffraction experiment fails. - Avoid submitting impure material. X-ray quality crystals should be grown purposely, and should not usually be derived from purification or characterization attempts (e.g. crystals coming from NMR tubes is discouraged).
- If crystals do not grow from one solvent, try another. Be aware that growing crystals from certain solvents may result in crystals containing disordered solvent, and this may impede the structure determination.
- Please ensure that the sample is sufficiently characterized by other methods so that its chemical formula is mostly correct. One of the most common mistakes is the submission of a material that is not what the chemist thinks it is.
- Devise a numbering scheme for the proposed molecule prior to sample submission, and include this numbering scheme with the submission form.
- Submitting air stable crystals in small sample vials is preferred, but not necessary. Samples submitted in round-bottomed flasks or other glassware that impedes optical examination and microscopic manipulation of the crystals are not preferred.Ê Air-sensitive materials can be submitted in any type of container.
- Evaporation of samples to dryness during recrystallization may result in the crystals adhering to the walls of the container. This may increase the likelihood that the crystals are crushed when the crystallographer attempts to isolate potential specimens.
Remember the old crystallographic saying: Garbage In = Garbage Out
FAQ
PXRD sample submission
Click here to print a Submission Form (Adobe PDF format).
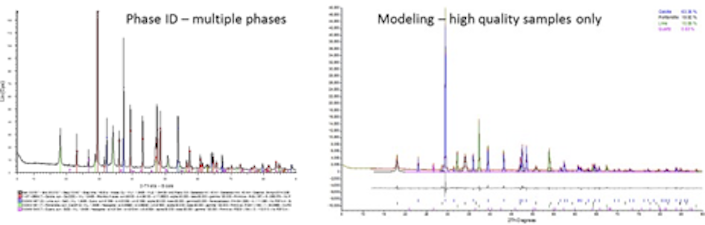
Powder or polycrystalline X-ray diffraction (PXRD) encompasses many applications that can be useful to the research and study of natural and new materials. While PXRD is most commonly used for phase identification and crystallographic investigations, there are a variety of PXRD methods and techniques available that can be useful to researchers across many different fields.
With several diffractometers available, this facility can accommodate different configurations required for use with specific methods. Please note that there are many techniques available but some require high quality samples and so not every technique can be applied to every sample.
Due to the complexity of managing different available configurations with a variety of measurement protocols and varying sample types, clients should contact the lab to discuss the best path forward. Client expertise is recommended or required in some cases.
A note to students
As a student, you have an opportunity to learn about a technique that is critical to revealing structural properties in materials. We have a variety of software programs available for you to use to process data and to develop your skills in crystallography. You should have a basic understanding of the following concepts before undertaking PXRD:
- the Bragg equation and diffraction
- unit cell and crystal systems
- crystalline vs amorphous
- crystallographic information file (CIF)
A number of links to resources with educational materials for XRD are shown on the links page.
Powder Diffraction (with 0D or 1D detectors)
We offer services that use conventional powder diffraction methods. These methods usually require the sample to be in powder form, randomly oriented, ideally 1-5 microns in size, and in sufficient quantity for best results. For samples that are not powders, cannot be made into a powder, or are limited, we may still be able to obtain valuable information about the material – we have done phase identification on some very challenging samples. Please contact us for more details.
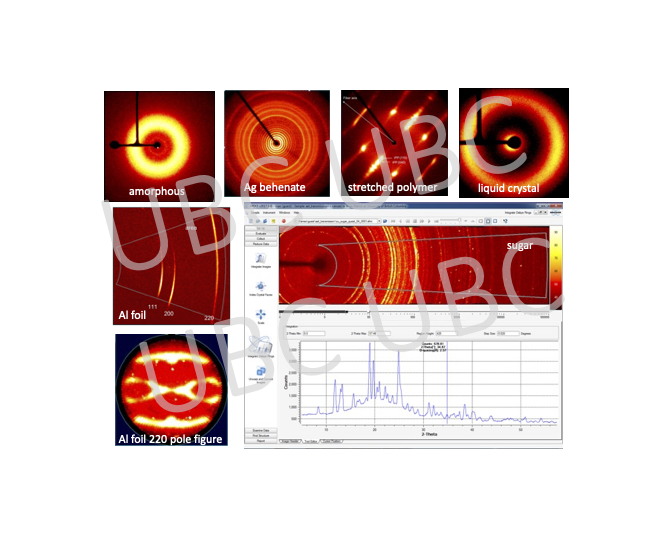
2D Diffraction
Traditionally, powder diffractometers have used point (0D) or linear (1D) detectors. Additionally, an area (2D) detector can be a useful tool to help reveal structural aspects of a material. We have 2D detectors on the single crystal instruments as well as on the Empyrean 3 multi-purpose diffractometer.
- Examples of sample types that can be used:
- Films or plastics for diffraction in transmission mode
- Capillaries for liquid crystals, suspensions, or air-sensitive samples
- Micro samples for micro-diffraction
- Precious samples for non-destructive testing
- Solid materials
- Gels
- Powder, polymer, and textured samples for checking graininess or orientation
If you have questions about our PXRD services please contact Anita Lam at 604-822-4865.
