
Elliott Burnell
Profile
Research and Teaching Interests
Nuclear magnetic resonance (NMR) has become an important technique for the study of molecular systems, and in recent years both liquid and solid type NMR experiments have been applied to ordered fluids, including liquid crystals, soaps, biological membranes, and solute molecules partially oriented in liquid-crystal solvents. Dr. Burnell's research exploits these NMR methods to investigate liquid crystalline systems.
The normal single-quantum proton NMR spectra of partially oriented molecules often contain hundreds of lines. For example, the lower spectrum is that of a mixture of p-xylene, p-dichlorobenzene and 1,3,5-trichlorobenzene dissolved in a nematic liquid crystal.
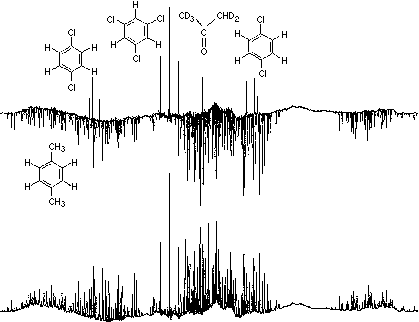 In the upper spectrum a computer fit to the p-xylene spectrum has been subtracted (negative peaks) leaving the simpler spectra of the other two solutes as positive peaks. Two-dimensional multiple-quantum NMR techniques were employed to make tractable the analysis of the very complex p-xylene single-quantum spectrum.
In the upper spectrum a computer fit to the p-xylene spectrum has been subtracted (negative peaks) leaving the simpler spectra of the other two solutes as positive peaks. Two-dimensional multiple-quantum NMR techniques were employed to make tractable the analysis of the very complex p-xylene single-quantum spectrum.
From such experiments much valuable molecular information, such as accurate molecular geometries and various anisotropic properties of the molecules, is obtainable. In particular, Dr. Burnell's group is interested in the average orientation of both the liquid crystal molecules and of the dissolved solutes, and in the intermolecular forces that are responsible for this orientational order. The group is studying these anisotropic intermolecular forces from two points of view. In one approach, they are using mean-field models to describe both long- and short-range forces. The dominant contributions are 1) short-range repulsive interactions and 2) interactions between the solute molecular quadrupole moment and the mean electric field gradient that is present in most liquid crystals. In the other approach, they are carrying out Monte Carlo computer simulations, which incorporate both short-range repulsive and electrostatic interactions, to model the liquid crystal solutions used in NMR studies. These computer simulations of NMR experimental results demonstrate an important feature of Dr. Burnell's approach to research, i.e. the interplay between experiment and theory.

Contact
Curriculum Vitae
B.Sc., Memorial (1965); M.Sc., Memorial (E. Bullock, 1968); Ph.D., Bristol (A.D. Buckingham, 1970); Postdoctoral, British Columbia (M. Bloom, 1969-71) and Basel (P. Diehl, 1971-72).
