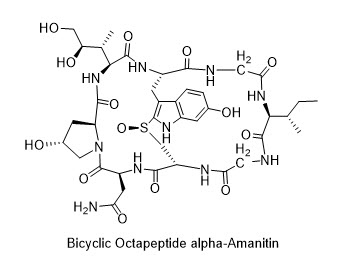
Abtract: Alpha-amanitin, one of deadliest toxins known to humankind (LD50 50-100 μg/kg), is a bicyclic octapeptide isolated from the notorious death-cap mushroom, Amanita phalloides. Isolated over sixty years ago, alpha-amanitin is a potent, heat-stable, orally available, highly selective, allosteric inhibitor of RNA polymerase II (Pol II). Its bicyclic octapeptide structure contains oxidized amino acids that are key to its toxicity: trans-hydroxy-proline (Hyp) and notably (2S,3R,4R)-4,5-dihydroxy-isoleucine (DHIle), along with a crosslink comprising 6-hydroxy-tryptathionine-(R)-sulfoxide that is unique among all natural products.
Alpha-amanitin kills both rapidly growing and quiescent cells by inhibiting Pol II, leading to apoptosis. Recently, it was shown that, when injected in sub-lethal doses, this toxin prevents cancer relapse in mice bearing tumor xenografts that are resistant to common chemotherapeutics.
As fermentation yields are low, a synthetic source of the toxin is urgently needed to prepare antibody drug conjugates (ADCs) for use in clinical oncology. That alpha-amanitin has resisted synthesis is reflected in three implicit challenges: i) a synthetic route to oxidatively-delicate 6-hydroxy-tryptathionine, ii) an enantioselective synthesis of (2S,3R,4R)-4,5-dihydroxy-isoleucine, and iii) a diastereoselective sulfoxidation to favor the R-sulfoxide.
Herein we report the first total synthesis of alpha-amanitin, which surmounts the key challanges mentioned above. The identity of the final synthesized toxin was confirmed by UV absorbance, mass spectrometry, HPLC co-injection of the synthesized and authentic alpha-amanitin, and finally the 1H-NMR of the obtained product, hence concluding the total synthesis.