
Yoshikata Koga
Profile
Research and Teaching Interests
Dr. Koga's group has introduced a new approach to the thermodynamic studies of aqueous solutions. Methods of measuring various thermodynamic quantities differentially were devised. Using these, the inter-molecular interaction, the key quantity in the so-called "many-body problem", can be evaluated experimentally.
For the aqueous solutions of 2-butoxyethanol, a common model system for solutions of biochemical importance, it was pointed out that there are regions in the composition-temperature field, in each of which the "mixing scheme" is qualitatively different. Furthermore, the transition from one mixing scheme to the other was found much more subtle than the normal phase transition. At the boundary separating two mixing schemes, the thermodynamic quantities that are proportional to the third derivatives of the Gibbs energy showed anomalies. (In contrast, the normal phase transitions are accompanied by anomalies in the second derivatives; heat capacity, compressibility etc.) The boundary separating the two regions has been dubbed as the "Koga linien" by Danish collaborators.
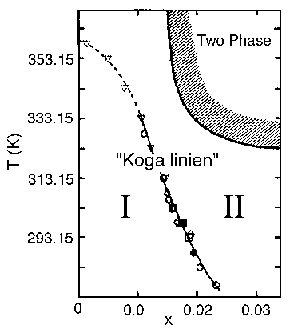
The group has started applying this new methodology to aqueous solutions of biopolymers. In particular, the effect of a third component on the function and the state of biopolymers in queous solutions is now studied.
There is a preliminary indication that "Koga linien" plays an important role in dictating the nature of biopolymers.
As a recognition of his contribution to solution thermodynamics, he was awarded the Society Award by The Japan Society of Calorimetry and Thermal Analysis in 2006.
He published a monograph, "Solution Thermodynamics and Its Application to Aqueous Solutions: A Differential Approach", Elsevier, Amsterdam (2007) ISBN:978-0-444-53073-8 summarizing his works up to about 2006.
In 2011, he obtained for the first time in the world a fourth derivative of Gibbes energy and named it "Accerelation of the effect of solute on entropy-volume cross fluctuation density." See J. Chem. Phys. 134(2011) 214502.
Contact
Curriculum Vitae
Senior Instructor Emeritus; B. Sc., Tokyo (1960); M. Sc.,Tokyo (1962); Ph. D., British Columbia(L. G. Harrison, 1969); Teaching Postdoctoral, British Columbia (L. G. Harrison 1969 -71);Visiting Scholar, McMaster (J. A. Morrison,1975, 76, 84, 86, 87); Guest Professor,Tokyo Institute of Technology (1993-94, 2000). Guest Professor, Technical University of Denmark (2000). Guest Professor, Chiba University (2001-2002, 2003, 2004). Guest Professor, Roskilde University (2003) , ”JSCTA Award” from the Japan Society of Calorimetry and Thermal Analysis (2006). Published monograph, "Solution Thermodynamics and Its Application to Aqueous Solutions: A Differential Approach." Elsevier, Amsterdam (2007). Obtained for the first time in the world a fourth derivative of Gibbes energy and named it "Accerelation of the effect of solute on entropy-volume cross fluctuation density." See J. Chem. Phys. 134(2011) 214502.
